Inorganic Chemistry Questions
The best high school and college tutors are just a click away, 24×7! Pick a subject, ask a question, and get a detailed, handwritten solution personalized for you in minutes. We cover Math, Physics, Chemistry & Biology.

Inorganic Chemistry
MetallurgyIn the coal-gasification process, carbon monoxide is converted to carbon dioxide via
the following reaction:
CO (g) + H2O (g) → CO2 (g) + H2 (g)
In an experiment, 0.35 mol of CO and 0.40 mol of H₂O were placed in a 1.00-L
reaction vessel. At equilibrium, there were 0.17 mol of CO remaining. Keq at the
temperature of the experiment is_____________.
5.5
1.2
1.0
0.75
0.87

Inorganic Chemistry
Preparation and Properties of CompoundsDraw the Lewis structure with the lowest formal charges for NO. Include any nonzero formal
charges and lone pair electrons in the structure.

Inorganic Chemistry
Preparation and Properties of CompoundsClick in the answer box to activate the palette.
The half-life of thallium-201 is three days. What fraction of thallium-201 is still present in an
individual after twelve days? (Report your answer in fraction format.)

Inorganic Chemistry
Preparation and Properties of CompoundsMatch the following scientists with their discover/belief.
-Earnest Rutherford
-Niels Bohr
-JJ Thomson
-Democritus
-Werner Heisenberg
-John Dalton
Used the quantum theory to predict that electrons orbit the nucleus of an atom
Proposed atoms are composed of positive and negative charges and believed
that electrons are evenly disbursed within the atom
Proposed different kinds of atoms called elements and stated that atoms cannot
be created or destroyed
Proposed all matter is composed of tiny, indivisible particles called atoms
Discovered the atom's positive charge was in the center (nucleus) of the atom
Believed the exact location of electrons at a given instance to be "uncertain"

Inorganic Chemistry
P Block - Group 16YOU MUST SUBMIT WORK FOR THIS PROBLEM.
Draw the Lewis structure for H₂CS. You must show adding up the valence electrons.
Determine the following for the structure:
•Molecular Shape
• Bond Angle
• Polarity

Inorganic Chemistry
Preparation and Properties of CompoundsYOU MUST SUBMIT WORK FOR THIS PROBLEM.
You are conducting Experiment 8: Molar Mass by Vapor Density as directed in the lab manual. You have determined the following. The volume of the flask is 255 mL. The boiling
water temperature is 98.9°C. The barometric pressure is 773.4 mm Hg
A) How will you determine the mass of your sample of gas?
B) If the mass is determined to be 0.630 g. what is the molar mass of the gas?
HELPFUL REFERENCE INFORMATION:
PV = nRT
atm L
R = 0.08206 atm L/mol K
1 atm = 760 mmHg
For the toolbar, press ALT+F10 (PC) or ALT+FN+F10 (Mac).

Inorganic Chemistry
HydrogenBe sure to answer all parts.
Write a balanced equation showing how this nucleus decays to form an a particle: Es-252.
(Give all nuclei in the form AZX.)

Inorganic Chemistry
Qualitative analysisIf the concentration of reactant X after 37s in a reaction was 0.0090 mol/L and after
80s was 0.0010 mol/L, calculate the average reaction rate.
Answer to 5 decimal places in mol/Ls. Leave out units

Inorganic Chemistry
Qualitative analysisUse the collision theory to provide an explanation of each of the following (2A each):
a. Food spoils more rapidly on a counter than in a refrigerator
b. Magnesium metal reacts much more quickly in concentrated HNO3 rather than dilute HNO3
c. Large piles of newspapers burn more quickly than the same mass of wood logs
d. Paints and stains often have instructions that they should not be applied below 10°C

Inorganic Chemistry
HydrogenArrange the following bonds by increasing bond polarity.
CI-P Cl-Ge CI-S CI-CI Cl-Br

Inorganic Chemistry
Preparation and Properties of CompoundsBe sure to answer all parts.
The acid in acid rain is generally sulfuric acid (H₂SO₂). When this rainwater falls on statues
composed of marble (CaCO3), the H₂SO4 slowly dissolves the CaCO3. Write a balanced equation for this acid-base reaction. (Include the phase of the substances.)

Inorganic Chemistry
Qualitative analysisIn electroplating of chromium, how many grams of chromium should be
electrodeposited if you pass 4.60 A current through a solution of 0.180 M CrCl3
for one hour? (atomic weights: Cr 52.00; Cl 35.45 amu)
A.2.98 g
B.3.12 g
C. 0.446 g
D.9.06 g
E.4.02 g

Inorganic Chemistry
Preparation and Properties of CompoundsIf the outside of the flask is not dried after vaporizing the liquid, will the molar mass of
the compound be too high or too low? Explain.

Inorganic Chemistry
Classification of Elements and PeriodicitySuppose you assumed that the atmospheric pressure was one atmosphere instead of the
pressure that you recorded. Describe the error that would occur in reporting the molar
mass.

Inorganic Chemistry
Qualitative analysisDetermine pH Solution containing 0.610 M HClO2 and 0.215 M LiClO2 if Ka of HClO2 is 6.30 x 10^-4.
A PH 3.78
B. PH 8.35
C.PH 3.65
D.PH 5.78
PH 2.75

Inorganic Chemistry
Qualitative analysisDetermine pH of solution containing 0.210 M C6H7NH3Cl
(Kb of C6H7NH2 is 3.50 x 10^-7)
A. PH 5.35
B, pH 4.67
C. PH 4.11
D.PH 10.9
E. PH 3.57

Inorganic Chemistry
P Block - Group 15When 5.5 g N₂ reacts with 2.2 g H₂ in the reaction shown below, how many grams of ammonia (NH3) will be produced? Show your work or explain your process.
N₂ + 3 H₂ --> 2 NH3

Inorganic Chemistry
Classification of Elements and Periodicity2. a) Draw the Lewis structure of sulfite (SO32-) with minimized formal charges on all atoms and assign
partial charges to each atom
b) How many electrons is around central S atom?
c) What is the hybridization of each atom?

Inorganic Chemistry
Qualitative analysisSince vanadium can have more than one possible oxidation state, there are several different forms of vanadium oxide. One form of vanadium oxide contains 56% V. What is the name and formula of this compound? Show your work or explain your process.

Inorganic Chemistry
Qualitative analysisVery dilute solutions can be measured in parts per billion that is, the number of parts in 1,000,000,000 parts of solution. To be effective, the concentration of digoxin, a drug used to treat congestive heart failure, must be 0.5-2.0 ng/mL. Convert both values to parts per billion (ppb).

Inorganic Chemistry
Qualitative analysisHow many milliliters of a 1.40 M glucose solution contains each of the following number of moles?
a. 0.0740 mol
b. 3.60 mol

Inorganic Chemistry
Qualitative analysisPropyl acetate (CsH1002; molecular weight = 102.13 g/mol) is a water-soluble ester with a characteristic pear-like odor. A lab technician dissolves 5 g of this ester in enough water to give 250 mL of solution. Calculate the molarity of this solution, showing ALL your work. (2 pts)

Inorganic Chemistry
Classification of Elements and PeriodicityDo atoms generally get bigger or smaller as you go down a group in the periodic table?

Inorganic Chemistry
Preparation and Properties of CompoundsWrite the full electron configurations for the following elements.
F
Mg2+
Si
N
P
S²-
K+
Ni
As
Kr

Inorganic Chemistry
Qualitative analysisThe initial volume of a gas cylinder is 750.0 mL. If the pressure of a gas inside the cylinder changes from to, what is the final volume the gas occupies?
O2.14 L
3.151 L
0.321 L
1.750 L
0.630 L

Inorganic Chemistry
Coordination compoundsIf you were writing an electron configuration for a bromine atom, which elemental symbol would you place in the
bracket?
Kr
Xe
Ar
Ne
He

Inorganic Chemistry
MetallurgyLattice energy is _____
the sum of ionization energies of the components in an ionic solid
the sum of electron affinities of the components in an ionic solid
the energy given off when gaseous ions combine to form one mole of an ionic solid
the energy required to convert a mole of ionic solid into its constituent ions in the gas phase
the energy required to produce one mole of an ionic compound from its constituent elements in their standard states

Inorganic Chemistry
Qualitative analysisGas density can be calculated by dividing the mass of gas by its volume. If you took a balloon of gas and then warmed the balloon in a sunny window, what can now be said about the density of the gas in the balloon?
The gas density will increase.
The gas density will remain the same.
The density of gases is independent of temperature.
The gas density will decrease.

Inorganic Chemistry
Classification of Elements and Periodicity"When filling orbitals of equal energy, electrons fill them singly first with parallel spins." This is known as:
Bohr's model.
Ground state.
Hund's rule.
Pauli exclusion principle.

Inorganic Chemistry
MetallurgyWhat is the wavelength (angstroms) of a photon that has an energy of 4.38 x 10^-18 J?
454 angstroms
1.31 x 10^9 angstroms
6.89 x 10^15 angstroms
1.45 x 10^-16 angstroms
2.30 x 10^7 angstroms

Inorganic Chemistry
MetallurgyThe greater the lattice energy, the greater the charges on the participatory ions and the smaller their radii.
True
False

Inorganic Chemistry
Preparation and Properties of CompoundsIn Experiment 4, you isolate iron(III) hydroxide from the following reaction:
Fe(NO3)3(aq) + 3 NaOH(aq) → Fe(OH)3(s) + 3 NaNO3(aq)
What method was used to isolate this product? Why was this method used?

Inorganic Chemistry
MetallurgyOf the following transitions in the Bohr hydrogen atom, the
n=7→n=3
n=4 →n=2
n=4 → n = 7
n=3→n=2
All transitions absorb plotons of equivalent energy.

Inorganic Chemistry
MetallurgyAn FM radio station broadcasts electromagnetic radiation at a frequency of 90.2 MHz. The wavelength of this radiation is
2.71 x 10^10
0.301
3.33
2.71 x 10^16
3.33 x 10^6

Inorganic Chemistry
MetallurgyElectromagnetic radiation with a wavelength of 665 nm appears as orange light to the human eye. The frequency of this light is
4.51 x 10^5
2.22 x 10^-15
4.51 x 10^14
2.00 x 10^11
2.00 × 10²

Inorganic Chemistry
Preparation and Properties of CompoundsDetermine the pH at the equivalence point of a titration between 50.0 mL of 0.173 M (CH3)2NH solution with 0.173 M HCIO4.

Inorganic Chemistry
Preparation and Properties of CompoundsIs the calculated sodium hydroxide concentration higher, lower or the same as the actual sodium hydroxide concentration in the flask, if the oxalic acid that was weighed was not completely transferred to the volumetric flask? Explain your answer.

Inorganic Chemistry
MetallurgyFor this question, Kryptonite (Kp) is a fictional transition metal with all possible
charges from +1 to +6. Name the following compound:
KpP2
For this question, Vibranium (Vb) is a fictional metal in group 2 of the Periodic table.
Write the formula for Vibranium Nitride

Inorganic Chemistry
MetallurgyIf you prepare a solution by adding sufficient amount of solute so that after heating and cooling the solution there is a visible amount of solid solute left in the bottom of the beaker, the solution would be considered _______
supersaturated
unsaturated
thermally saturated
saturated

Inorganic Chemistry
MetallurgyFor this question, Adamantium (A) is a fictional transition metal with multiple possible charges.
Write the formula for: Adamantium (II) Hyponitrite

Inorganic Chemistry
Preparation and Properties of CompoundsA student prepares a dilute solution of sodium hydroxide, NaOH(aq), starting with 6 M so- dium hydroxide. She then titrates a 1.372 g sample of KHP with the dilute sodium hydroxide solution, NaOH(aq), to a phenolphthalein end point. If the titration required 21.84 mL of sodium hydroxide, NaOH(aq), calculate the molar concentration of the sodium hydroxide solution, NaOH(aq). (Remember that KHP is potassium hydrogen phthalate, KHC,H,O, NOT potassium hydrogen phosphorus!)

Inorganic Chemistry
Classification of Elements and PeriodicityUse what you just observed to predict the following. How many drops of water can you put onto a penny before the water spills over the side? How many drops of alcohol before the alcohol spills over the side. Make a prediction, give it a try, and, most importantly, explain what's happening.
Inorganic Chemistry
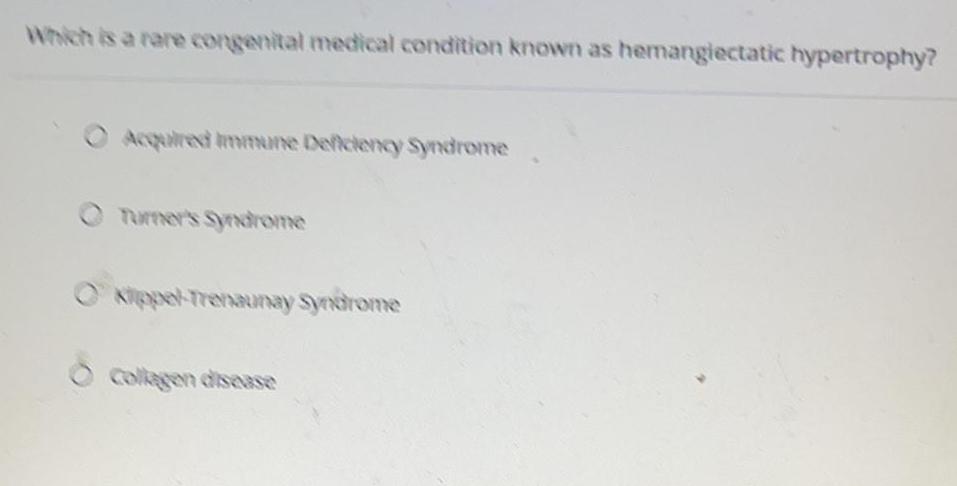
Qualitative analysisWhich is a rare congenital medical condition known as hemangiectatic hypertrophy?
Acquired Immune Deficiency Syndrome
Turner's Syndrome
Okippel-Trenaunay Syndrome
Collagen disease

Inorganic Chemistry
Classification of Elements and PeriodicityWrite the complete electron configuration for the vanadium atom.
Using NOBLE GAS notation write the electron configuration for the copper atom.

Inorganic Chemistry
Preparation and Properties of CompoundsWhich of the following statements is/are true for a compound AX4, where A has 6 valence electrons
and X has 7 valence electrons?
a) The Lewis structure cannot be drawn without violating the octet rule.
b) The central atom, A, has 4 bonding and 2 non-bonding electron pairs around it.
c) In order to create a valid Lewis structure, it must contain at least one double bond.

Inorganic Chemistry
Preparation and Properties of CompoundsWhat is the total number of moles of products involved in the following reaction?
CaCO3 (s) + 2HCI (aq) → CaCl₂ (aq) + CO₂ (g) + H₂O(g)
6
2
3
5

Inorganic Chemistry
Qualitative analysisGiven the following information, what is the ΔHºf (heat of formation for ZnS(s)?
Thermochemical equation: 2ZnS(s) + 302(g) → 2ZnO(s) + 2SO2(g)
ΔH°f of ZnO(s) = -348.3 kJ/mole
ΔH°f of SO2(g) = -296.8 kJ/mole
The heat of formation is
ΔH°rxn = -878.2 kJ

Inorganic Chemistry
Qualitative analysisWhat is the change in enthalpy in joules when 5.44 x 10^-4 mol of AgCl dissolves in water according to the following chemical equation:
AgCl(s)→ Ag (aq) + CI (aq) ΔH = 65.5 kJ

Inorganic Chemistry
MetallurgyRelate how intensity is different than texture
Intensity is how Intense a plece of music Is and the texture is how hard or soft it sounds.
Intensity is how dynamics, articulation and other factors that a composer may include in music affects the listener while texture is the amount of voices and how they work in cooperation with each other in a
piece of music.
Intensity is how hard or soft a piece of music sounds and texture is how intense it is.
Intensity is how dynamics, articulation and other factors that a composer may include in music affects the musicians playing it while texture is the amount of instr and how they work in cooperation with each other in a plece of music.

Inorganic Chemistry
Qualitative analysisWhat is the mass of one uranium pellet?
7 grams
70 grams
500 grams
50 grams