General Questions and Answers

Physical Chemistry
General1 A mixture of 2 3 g formic acid and 4 5 g oxalic acid is treated with conc H SO4 The evolved gaseous mixture is passed through KOH pellets Weight in g of the remaining product at STP will be a 1 4 c 2 8 b 3 0 d 4 4 2018 6

Physical Chemistry
GeneralThermodynamics 23 The heat of neutralization of LiOH and HCI at 25 C in 34 868 kJ mol The heat of ionisation of LiOH will be 1 44 674 kJ 2 22 232 kJ 3 32 684 kJ


Physical Chemistry
General1 pH of 10 8 M NaOH is a 8 0 b 6 0 c 6 98 d 2 Milliequivalent of Ba OH 2 present in 100 mL solution to have pH 13 is a 10 2 d 0 01 c 0 1 temperature under high pressure 10 10 b 10 At a to h

Physical Chemistry
GeneralThe heat of combustion of a reaction does not depend on a Physical state of the substance b Temperature at which combustion is carried out c Amount of oxygen present d Whether the combustion is carried out

Physical Chemistry
General6 The equation for the formation of lithium bromide is Li Br LiBr If 5 0 grams of each reactant are used Which substance is the limiting reagent F Lithium G Bromine H Lithium bromide 1 There is no limiting reagent

Physical Chemistry
General3 If sulfur has two isotopes one with a mass of 32 and the other with a mass of 35 and the average mass is 32 1 if you pulled out a single sulfur atom would it have a mass of 32 1 Why or why not

Physical Chemistry
General4 Varnish is a homogeneous colloidal dispersion solution of in oil or thinne or in both apreenshot A Pigment B Resin C Salt D Gypsum 5 The material which are used to prevent the flow of heat are known as A Insulators B Conductors C Semiconductor D Paints cty

Physical Chemistry
GeneralAcetic anhydride is prepared in the laboratory by heating sodium acetate with 1 Ethyl chloride 2 Acetyl chloride 3 Conc H SO 4 Zinc dust An unknown alcohol is treated with the Lucas reagent to determine whether the alcohol is primary secondary or tertiary Which alcohol reacts fastest and by what mechanism 1 Secondary alcohol by S 2 2 Tertiary alcohol by S 2 4 Tertiary alcohol by S 1 3 Secondary alcohol by S 1

Physical Chemistry
GeneralBoard Competitive Exams 44 Change in enthalpy for reaction 2H O 2H O 1 O g if heat of formation of H O and H O l are 188 and 286 kJ mol respectively is 1 196 kJ mol 2 196 kJ mol 3 948 kJ mol 049

Physical Chemistry
GeneralIn order to oxidise a mixture of one mole of each of FeC O4 Fe C O4 3 FeSO4 and Fe SO4 3 in acidic medium the number of moles of KMnO4 required is JEE Main 2019 8 April Shift 1 c 3 d 1 5 a 2 b 1

Physical Chemistry
GeneralTo assess the stoichiometry of a reaction one must use the Select the correct answer below Ounbalanced chemical equation O balanced chemical equation O masses of the substances O volumes of the substances

Physical Chemistry
GeneralCalculate the ratio of solubility of AgCl in 0 1M AgNO and in pure water Given K of AgCl 1 8 x 10 0 1 1 34 x 10 5 M 3 2 x 10 8 M 2 1 34 x 10 4 M 4 3 x 10 6 M

Physical Chemistry
GeneralWhich of the following statements is are INCORRECT All spectral lines belonging to Balmer series in hydrogen spectrum lie in visible region If a light of frequency v falls on a metal surface having work function hvo photoelectric effect will take place only if vs Vo The number of photoelectrons ejected from a metal surface in photoelectric effect depends upon the frequency of incident radiations 4 The series limit wavelength of Balmer series of H atom is R where R is Rydberg s constant

Physical Chemistry
General21 For the gas phase reaction PCI 9 PCL g Cl g which of the following conditions is correct AIPMT Prelims 2008 1 AH 0 and AS 0 2 AH 0 and AS 0 3 AH 0 and AS 0 4 AH0 and AS 0

Physical Chemistry
General4 DCM and H O will make turbid colloidal mixture Hard water can block radiators due to the formation of 1 Insoluble calcium and Magnesium salts 3 Insoluble Phosphate salts 2 Insoluble Sodium salts 4 Insoluble Potassium salts Which one of the two lodine atoms will be more reactive in the SN and SN reaction

Physical Chemistry
GeneralWrite a short story in about 100 120 on the basis of the following inputs given in th old lady alone maid left two robbers in early 20s robbery woman stabbed in hospital assailants expected to be caught

Physical Chemistry
GeneralWhich of the following statement is incorrect about silicones O R SiCl RSiCl3 are monomers of silicones O Silicones are hydrophobic O R3SiCl is used to close polymeric chain O Polymers of SiO4


Physical Chemistry
GeneralA permanganate solution is prepared by dissolving 20 0123 g KMnO4 in 500 mL of distille water and boiled for 1 hour to remove any organic material Following sintered glas filtration the solution is quantitatively transferred to a 1 0 L volumetric flask and diluted volume with distilled water The permanganate solution was titrated against 0 1023 oxalic acid prepared in sulfuric acid solution A 50 00 mL aliquot of oxalic acid solut required 16 68 mL of permanganate solution A titration blank required 0 04 mL permanganate What is the permanganate molarity 5 H C O4 2 MnO4 6H S 10 CO 2 Mn 8 H O

Physical Chemistry
General22 1 g sample of chalk is completely neutralised by 100 ml 0 04N HCI solution w w CaCO3 in the sample is are 1 20 3 60 2 40 4 80

Physical Chemistry
GeneralProblem 1 1 Mass of an atom of oxygen in gram is 26 56896 x 10 24 g What is the atomic mass of oxygen in u

Physical Chemistry
GeneralChoose the disproportionation reaction from the following a XeF4 H O b Cl NaOH c P4 NaOH aq d XeF2 H O O a b Ob c O b c and d O a b c

Physical Chemistry
General3 Insoluble Phosphate salts 4 Insoluble Potassium salts Which one of the two lodine atoms will be more reactive in the SN and SN reaction 1 A will be faster in SN reaction but slower in SN 2 A will be faster both in SN and SN reaction 3 A and B will be equally reactive 4 will be factor in both SN and SN reaction

Physical Chemistry
Generala 12 4 D 140 Which of the following would increase the solubility of Pb OH 2 a Add hydrochloric acid b Add a solution of Pb NO3 2 c Add a solution of NaOH d None of the above the solubility of a compound is constant at constant temperat

Physical Chemistry
GeneralQuestion 4 The value of kp for the reaction at 27 C Br e Cl g 2BrCl g is 1 atm At equilibrium in a closed container partial pressure of BrCl gas is 0 1 atm and at this temperature the vapour pressure of Br2 e is also 0 1 atm Then what will be minimum moles of Br e to be added to 1 mole of Cl initially to get above equilibrium situation Options 10 6 a moles b moles 15 Vedantu YOUR PERSONAL TEACHER ONL
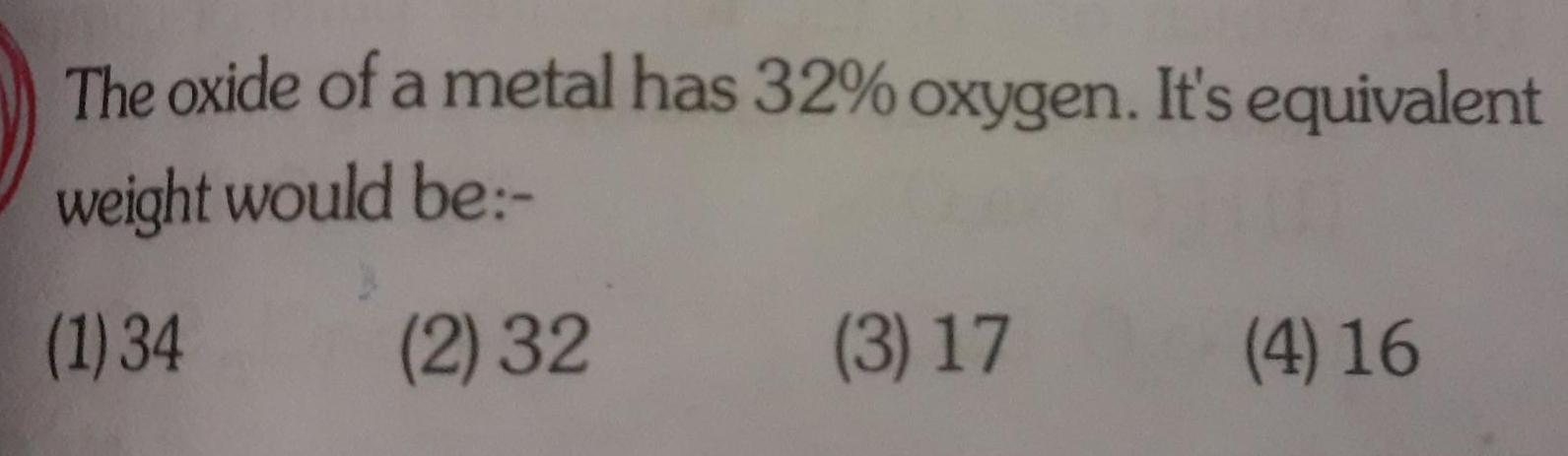
Physical Chemistry
GeneralThe oxide of a metal has 32 oxygen It s equivalent weight would be 1 34 2 32 3 17 4 16

Physical Chemistry
GeneralAmongst the compounds given the one that would form a brilliant coloured dye on treatment with NaNO in dil HCI followed by addition to an alkaline solution of B naphthol is 1 N CH 2 NHCH 3 H C NH CH NH

Physical Chemistry
GeneralA catalyst cannot affect A Product B Rate of reaction C Reactant D Both A B The lattice site in a pure crystal cannot be occupied by A Molecule B Ion FI

Physical Chemistry
GeneralThe enthalpy of neutralisation of HCI and NaOH is 57 kJ mol The heat evolved at constant pressure in kJ when 0 5 mole of H SO4 react with 0 75 mole of NaOH is equal to O42 75 52 50 20 50

Physical Chemistry
GeneralThe weight of 350 ml of a diatomic gas at 0 C and 1 atm pressure is lg The weight of one atom is in gram Assume gases are ideal A 16 NA B 32 NA C 16NA D 32NA

Physical Chemistry
General34 The vapour density of a mixture of NO and N O4 is 39 at 25 C What is the mass of NO present in 100 g of the mixture mol 2 8 4 960 60 1 17 9 g g 2 222 4 66 4 g

Physical Chemistry
Generalc Al SO4 3 14 If Ksp BaSO4 is 1 1x10 10 then in which of the following cases BaSO4 is precipitate out a 100 mL of 4 x 10 M BaCl 300 mL of 6 0 x 10 4 M Na2SO4 b 100 mL of 4 x 10 4M BaCl 300 mL of 6 0 10 M Na2SO4 c 300 mL of 4 x 10 M BaCl 100 mL of 6 0 x 10 M Na SO4 d All of the abou

Physical Chemistry
GeneralThe enthalpy of fusion of water is 1 435 kcal m The molar entropy change for the melting of ice C is AIPMT Prelims 201 1 5 260 cal mol K 2 0 526 cal mol K 3 10 52 cal mol K 4 21 04 cal mol K

Physical Chemistry
GeneralQ 7 Which of the following coordination entity should be expected to absorb light of lowest frequency Cr en CrCl6 Cr NH3 Incorrect 1 002 4 Cr CN

Physical Chemistry
GeneralWhich of the following vitamin has isoprene units in its structure A Vitamin A B Vitamin C C Vitamin B
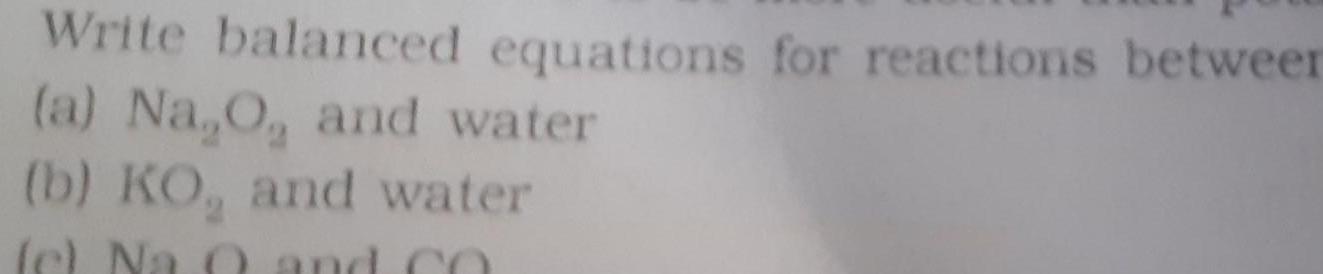
Physical Chemistry
GeneralWrite balanced equations for reactions between a Na O and water b KO and water c Na 0 and CO

Physical Chemistry
GeneralGiven that i 2C s 20 g 2CO g AH 786 4 kJ ii H g 1 2 O g H O l AH 285 8 kJ iii C H g O2 g 2CO g H O l AH 1297 kJ The enthalpy of formation of ethyne C H O 224 8 kJ O 224 8 kJ O 1072 2 kJ O 571 6 kJ

Physical Chemistry
GeneralThe inversion of cane sugar proceeds with half life of 500 minutes at pH 5 for any concentration of sugar However if pH 6 the half life changes to 50 minutes The rate law expression for the sugar inversion can be written as 1 r k Sugar H 6 2 r k Sugar H 3 r K Sugar H 1 4 r K Sugar H

Physical Chemistry
Generalev 5 The energy of an electron moving in nth Bohr s orbit of an element is given by En n eV atom Z atomic number The graph of E vs Z2 keeping n constant will be Z E gros Z O w Z d E 13 6 72 Z

Physical Chemistry
GeneralIn a gaseous reaction of the type aA bB cC dD which statement is wrong 1 a litre of A combines with b litre of B to giv C and D 2 a mole of A combines with b moles of B to giv C and D 3 g of A combines with b g of B to gi C and D 4 a molecules of A combines with b molecules B to giv Cand D R

Physical Chemistry
GeneralSECTION I STRAIGHT OBJECTIVE TYPE P N and H are taken in 1 3 molar ratio in a closed vessel to attain the following equilibrium N g 3H g 2NH g Find k for reaction at total pressure of 2P if PN at equilibrium is A 3P 8 1 B 3p C 4p 3 D None

Physical Chemistry
General9 At 20 C 0 258 mol A g and 0 592 mol B g are mixed in a closed vessel of 5 L capacity to conduct the following reaction A g 2B g C g If 0 035 mol C g remains in the equilibrium mixture then determine the partial pressure of each constituent at equilibrium Ans According to the equation 1 mol A g reacts with 2 mol B g to produce 1 mol C g Hence 0 035 mol A g and 2 0 035 0 07 mol B g are required to produce 0 035 mol C g Therefore equilibrium molar concentrations of different constituents will be as follows No of moles at equilibrium A g 0 258 0 035 0 223 2B g 0 592 0 07 0 522 C g 0 035 0 223 Equilibrium conc mol L 0 0446 As given T 273 20 K 293 K At equilibrium PA A RT 0 0446 0 0821 293 1 072 atm PB B RT 0 1044 0 0821 293 2 511 atm Pc C RT 7 x 10 3 x 0 0821 293 0 168 0 522 5 0 1044 0 035 7 x 10 3

Physical Chemistry
GeneralOn diluting the solution of an electrolyte a both and k increase b both and k decrease c A increases and k decreases d A decreases and k increases

Physical Chemistry
General67 Consider the reaction 2A B 3CP 20 Starting with 3 mole of A 2 mole of B and 6 mole of C number of moles of the products P and Q would respectively be a 2 and 4 b 4 and 2 c 3 and 1 5 d 1 5 and3

Physical Chemistry
General27 Specific volume of cylindrical virus particle is 6 02 x 10 cc g whose radius and length are 7 and 10 respectively If N 6 02 x 1023 find molecular weight of virus a 15 4 kg mol b 1 54 x 10 kg mol c 3 08 x 10 kg mol d 3 08 x 10 kg mol 2001

Physical Chemistry
GeneralGive examples a Positive radicals b Basic radicals c Composite radicals d Metals with variable valenc e Bivalent acidic radicals f Trivalent basic radicals

Physical Chemistry
GeneralThe crystalline salt Na2SO4 xH O on heating losses 55 9 of its mass The formula of crystalline salt is A Na SO4 5H O C Na2SO4 3H O B Na SO4 7H O D Na SO4 10H O

Physical Chemistry
General5 The practice of looking down upon the Blacks is known as a religions c racial discrimination b racial law d imperialism

Physical Chemistry
GeneralIf 10 17 J of light energy is needed by the interio of human eye to see an object The photons o green light 2 550nm needed to see the objec are A 27 B 28 C 29 D 30