Question:
H₂SO4(aq) + Pb(C₂H3O2)2(aq)-> PbSO4(s) + 2 HC₂H3O2(aq)
Last updated: 7/6/2022
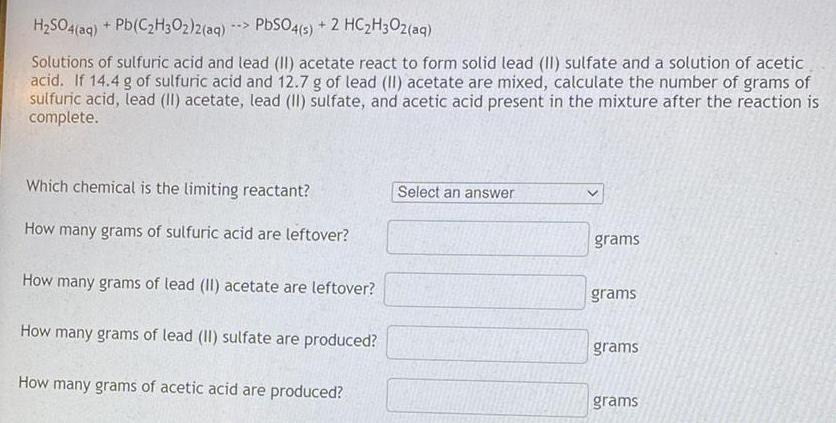
H₂SO4(aq) + Pb(C₂H3O2)2(aq)-> PbSO4(s) + 2 HC₂H3O2(aq) Solutions of sulfuric acid and lead (II) acetate react to form solid lead (II) sulfate and a solution of acetic acid. If 14.4 g of sulfuric acid and 12.7 g of lead (II) acetate are mixed, calculate the number of grams of sulfuric acid, lead (II) acetate, lead (II) sulfate, and acetic acid present in the mixture after the reaction is complete. Which chemical is the limiting reactant? How many grams of sulfuric acid are leftover? How many grams of lead (II) acetate are leftover? How many grams of lead (II) sulfate are produced? How many grams of acetic acid are produced?