Question:
The standard enthalpy of formation of NO2(g) is 33.1 kJ/mol.
Last updated: 7/7/2022
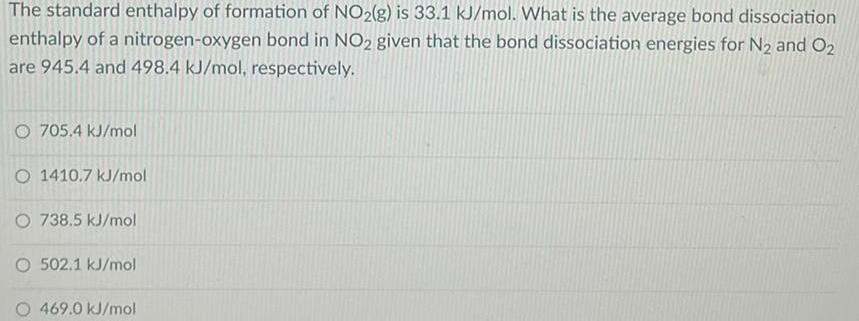
The standard enthalpy of formation of NO2(g) is 33.1 kJ/mol. What is the average bond dissociation enthalpy of a nitrogen-oxygen bond in NO2 given that the bond dissociation energies for N₂ and O₂ are 945.4 and 498.4 kJ/mol, respectively. 705.4 kJ/mol 1410.7 kJ/mol 738.5 kJ/mol 502,1 kJ/mol 469.0 kJ/mol