Question:
You are showing your friend how to do a molar conversion
Last updated: 7/5/2022
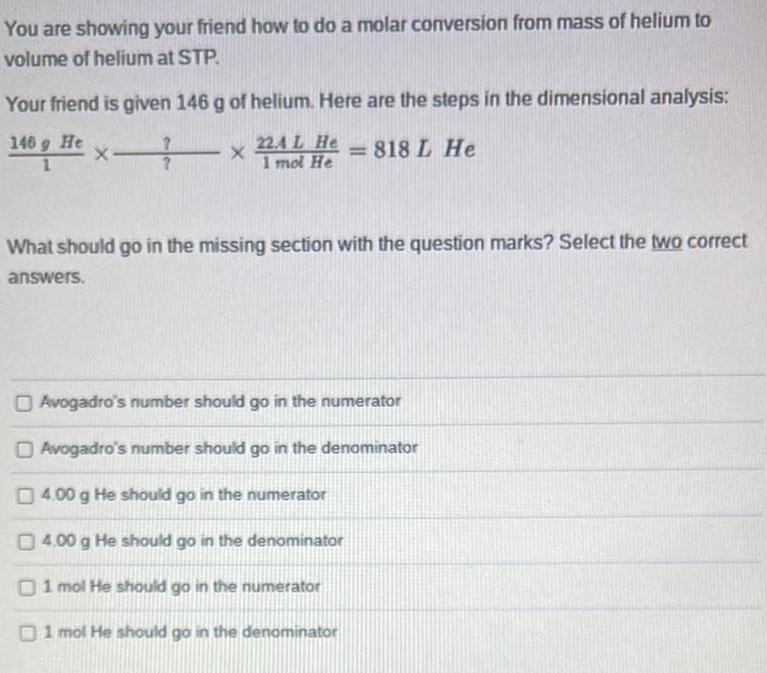
You are showing your friend how to do a molar conversion from mass of helium to volume of helium at STP. Your friend is given 146 g of helium. Here are the steps in the dimensional analysis: Avogadro's number should go in the numerator Avogadro's number should go in the denominator 4.00 g He should go in the numerator 4.00 g He should go in the denominator 1 mol He should go in the numerator 1 mol He should go in the denominator