Preparation and Properties of Compounds Questions and Answers

Inorganic Chemistry
Preparation and Properties of CompoundsThe gasoline in an automobile gas tank has a mass of 60.0 kg and a density of 0.752 g/mL
A) What's the specific gravity
B) Calculate the volume of gasoline in gallons (Show your calculations):

Inorganic Chemistry
Preparation and Properties of CompoundsSnO₂²- + 2NH₂OH = SnO3²- + N₂H4+ H₂O
In the above reaction, the oxidation state of tin changes from
How many electrons are transferred in the reaction?

Inorganic Chemistry
Preparation and Properties of CompoundsWhat volume of 2.5 M Nitric acid (HNO3) is required to prepare 500 mL of a 0.20 M HNO3 solution
40 mL
0.2 mL
500 mL
2.5 mL

Inorganic Chemistry
Preparation and Properties of CompoundsDoes a reaction occur when aqueous solutions of iron(II) iodide and lead(II) nitrate are combined?
no yes
If a reaction does occur, write the net ionic equation.
Use the solubility rules provided in the OWL Preparation Page to determine the solubility of compounds.
Be sure to specify states such as (aq) or (s).
If a box is not needed leave it blank.

Inorganic Chemistry
Preparation and Properties of CompoundsWhen the following skeletal equation is balanced under basic conditions, what are the coefficients of the species shown?
I2 + Cl- ---> I- + Cl2
Water appears in the balanced equation as a (reactant, product, neither) with a coefficient of (Enter 0 for neither.)
Which species is the oxidizing agent?

Inorganic Chemistry
Preparation and Properties of CompoundsIron(III) sulfide reacts with HCI (g) to produce iron(III) chloride and hydrogen sulfide. When 56.8 g of iron(III) sulfide reacts with 42.3 g of hydrochloric acid to produce 54.2 g of iron(III) chloride, what is the percent yield for
the reaction?
Molar masses:
Fe (55.85 g/mol)
S (32.07 g/mol)
Cl (35.45 g/mol)
H (1.008 g/mol)
Do NOT include units in your answer. If you round during your calculation, be sure to keep at least three (3) decimal places. Report your answer to one (1) decimal place.

Inorganic Chemistry
Preparation and Properties of CompoundsWhen the following half reaction is balanced under acidic conditions, what are the coefficients of the species shown?
Mn2+ H₂O ---> MnO₂ + H+
In the above half reaction, the oxidation state of manganese changes from _ to _

Inorganic Chemistry
Preparation and Properties of Compoundsto access important values if needed for this question.
When the following skeletal equation is balanced under basic conditions, what are the coefficients of the species shown?
Br₂ NH₂OH = N2H4+ Br2
Water appears in the balanced equation as a0 for neither.)
Which species is the reducing agent?

Inorganic Chemistry
Preparation and Properties of CompoundsA solution contains 35 g of KBr dissolvedin 205 g of water. Express the
concentration of the solution as %
(m/m).
14.6 % (m/m)
12.3 % (m/m)
5.86 % (m/m)
17.1 % (m/m)

Inorganic Chemistry
Preparation and Properties of CompoundsYou are exposed to a small amount of
radiation daily. This small radiation
exposure is the
background radiation
relative biological efficiency
personal radiation
inherent radiation

Inorganic Chemistry
Preparation and Properties of CompoundsAn unknown compound has the following chemical formula:
P₂Ox
where x stands for a whole number.
Measurements also show that a certain sample of the unknown compound contains 6.3 mol of oxygen and 2.50 mol of phosphorus.
Write the complete chemical formula for the unknown compound.

Inorganic Chemistry
Preparation and Properties of CompoundsCalculate the mass percent of element A in a compound with molecular
formula A2B3. The molar mass of element A is 35.5, and molar mass of element B is
48.5. When input answer, convert percentage to decimal. Use 3 decimal places. For
example, if you answer is 56.7%, convert it to 0.567

Inorganic Chemistry
Preparation and Properties of CompoundsThe movie below shows some molecules in a tiny sample of a mixture of gases.
Drag the slider to see how the molecules move.
Does a chemical reaction happen during this
movie?
If a chemical reaction does happen, write a
balanced chemical equation for it.

Inorganic Chemistry
Preparation and Properties of CompoundsMagnesium metal, Mg (s), reacts with hydrochloric acid to form aqueous magnesium chloride and hydrogen gas. When 43.5 g of magnesium is added to 258 mL of 6.00 M hydrochloric acid, what mass (in grams) of hydrogen is produced, assuming the reaction goes to completion?
Molar masses:
Mg (24.31 g/mol)
CI (35.45 g/mol)
H (1.008 g/mol)
Do NOT include units in your answer. If you round during your calculation, be sure to keep at least three (3) decimal places. Report your answer to one (1) decimal places.

Inorganic Chemistry
Preparation and Properties of CompoundsShow the correct link to describe the type of solution:
Sugar completely dissolves whenadded to hot coffee
A layer of sugar forms on thebottom of a glass of iced tea
Sodium chloride crystals do notdissolve in solution

Inorganic Chemistry
Preparation and Properties of CompoundsWhat is the oxidation number of bromine as a reactant and in the product?
Br in Br₂
Br in Al₂Br6

Inorganic Chemistry
Preparation and Properties of CompoundsDraw the Lewis structure for BF3 in the window below and then answer the questions that follow.
• Do not include overall ion charges or formal charges in your drawing.
/

Inorganic Chemistry
Preparation and Properties of CompoundsSulfur dioxide gas and liquid water are formed by the decomposition of aqueous sulfurous acid (H₂SO3).
Write a balanced chemical equation for this reaction.

Inorganic Chemistry
Preparation and Properties of CompoundsA chemistry student weighs out 0.0576 g of formic acid (HCHO₂) into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.1900 M NaOH solution.
'Calculate the volume of NaOH solution the student will need to add to reach the equivalence point. Round your answer to 3 significant digits.

Inorganic Chemistry
Preparation and Properties of CompoundsFor the following electron-transfer reaction:
Pb²+ (aq) + Zn(s) → Pb(s) +Zn²+ (aq)
The reduction half-reaction is:

Inorganic Chemistry
Preparation and Properties of CompoundsThe reaction represented by the equation:
C(s) + H₂O(1) CO(g) + H2(g)
was carried out at 27°C. If the enthalpy change was 4500 J and the
entropy change was 12 J. What was the free energy change AG?

Inorganic Chemistry
Preparation and Properties of CompoundsIf you wanted to prepare 50.0 mL of hydrogen, collected over water at 25°C on a day when the barometric pressure was 730 torr, what mass of aluminum would you react with a hydrochloric acid? (Balance the equation first.
Al(s) + HCi(aq)+H2(g)

Inorganic Chemistry
Preparation and Properties of CompoundsFor each of the following equilibria, write the expression for the equilibrium constant K. and state their units.
1. 2NO2 ---> N2O4
2. N₂ + 3H₂ ---> 2NH3
3. CH3CH₂CO₂H(1 ---> CH3CH₂OH(CH3CH₂CO₂CH₂CH30) + H₂O
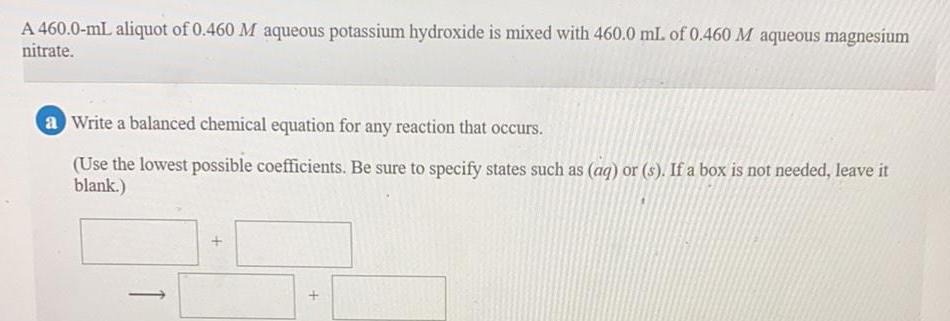
Inorganic Chemistry
Preparation and Properties of CompoundsA 460.0-mL aliquot of 0.460 M aqueous potassium hydroxide is mixed with 460.0 mL. of 0.460 M aqueous magnesium nitrate.
a Write a balanced chemical equation for any reaction that occurs.
(Use the lowest possible coefficients. Be sure to specify states such as (aq) or (s). If a box is not needed, leave it blank.)

Inorganic Chemistry
Preparation and Properties of CompoundsSodium hydrogen carbonate (NaHCO3), also known as sodium bicarbonate or "baking soda", can be used to relieve acid indigestion. Acid
indigestion is the burning sensation you get in your stomach when it contains too much hydrochloric acid (HCI), which the stomach
secretes to help digest food. Drinking a glass of water containing dissolved NaHCO3 neutralizes excess HCl through this reaction:
HCl(aq) + NaHCO3(aq) -> NaCl(aq) + H₂O(l) + CO₂(g)
The CO₂ gas produced is what makes you burp after drinking the solution.
Suppose the fluid in the stomach of a woman suffering from indigestion can be considered to be 150. mL of a 0.021 M HCl solution. What
mass of NaHCO3 would she need to ingest to neutralize this much HCl ? Round your answer to 2 significant digits.

Inorganic Chemistry
Preparation and Properties of CompoundsThe equation for the reaction of AgNO3
with Cu metal is given below. What mass
of Ag (s) will form from the reaction of
2.51 g of Cu, assuming that the Cu
reacts completely?
2AgNO3(aq) + Cu (s) → Cu(NO3)2 (aq) + 2 Ag (s)
0.466 g
4.26 g
8.52 g
0.739 g

Inorganic Chemistry
Preparation and Properties of Compounds2 K(s) + 2 H2O(1) 2 KOH(aq) + H2(g)
ΔH= ?
When 0.05 mol of K is added to 100 g of water, the temperature of the resulting solution rises from 25.00°C to 35.75°C. If the specific heat of the solution is 4.18 J/(g°C), calculate AH for the reaction.
A. None of these choices is correct
B.-5.41 kJ
C.-180 kJ
D.-360 kJ
E. -90 kJ


Inorganic Chemistry
Preparation and Properties of CompoundsConsider the reactions
1. C(graphite) + O2(g) →> CO2(g) ΔHxn=-393.5 kJ
2. H2(g) + 1/2O2(g) -> H₂O(1) ΔHOrxn=-285.8 kJ
3. 2 C2H2(g) + 5 O2(g) -> 4 CO2(g) + 2 H₂O0) ΔHºrn = -2598.8 kJ
Calculate the ΔHOrxn for the reaction:
2 C(graphite) + H2(g) -> C2H2(g) ΔHᵒrn = ???

Inorganic Chemistry
Preparation and Properties of CompoundsThe chemical formula for beryllium sulfide is BeS.
A chemist determined by measurements that 0.0200 moles of beryllium sulfide participate in a chemical reaction. Calculate the mass of beryllium sulfide that participates.
Round your answer to 3 significant digits.

Inorganic Chemistry
Preparation and Properties of CompoundsA chemist determined by measurements that 0.040 moles of potassium participated in a chemical reaction. Calculate the mass of potassium that participated in the chemical reaction.
Round your answer to 2 significant digits.

Inorganic Chemistry
Preparation and Properties of CompoundsBalance the chemical equation below using the smallest possible whole number stoichiometric coefficients.
C(s) + H₂(g) → C₂H6 (g)

Inorganic Chemistry
Preparation and Properties of CompoundsThe chemical formula for cesium chloride is CsCl.
A chemist measured the amount of cesium chloride produced during an experiment. She finds that 97.9 g of cesium chloride is produced. Calculate the number of moles of cesium chloride produced.
Round your answer to 3 significant digits.

Inorganic Chemistry
Preparation and Properties of CompoundsWhile Bayer is often credited with the commercialization of Aspirin (an chemical modification of a compound found in the Willow Tree bark) as a pain reliever medication. Indigenous Americans actually discovered the pain relieving properties of the Willow Tree long before Bayer. A compound extracted from Willow Bark was subjected to Elemental Analysis and the following mass data were collected:
C: 60.87%
H: 4.38%
O: 34.75%
Based on this information, what is the empirical formula of this compound?

Inorganic Chemistry
Preparation and Properties of CompoundsWhat is the balanced equation for the combustion of pentane when
the equation is balanced with the smallest, whole numbers
possible?
2 C5H12 +11 O2→ 10 CO + 12 H2O
C5H12 +8O₂ --> 5 CO₂ + 6H₂O
C5H1₂ →5C+ 6H₂
C5H12+5O2 → 5CO2+6H2
I DON'T KNOW YET

Inorganic Chemistry
Preparation and Properties of CompoundsWhat is the correct structure for 3-fluoro-2,2-dimethylpentane?

Inorganic Chemistry
Preparation and Properties of CompoundsThis is the chemical formula for zinc bromate:
Zn(BrO3)₂
Calculate the mass percent of oxygen in zinc bromate. Round your answer to the nearest percentage.

Inorganic Chemistry
Preparation and Properties of Compounds100.0 mL of 1.35 M HCI solution is mixed with 650.0 mL of distilled water. What is the final solution's concentration?
Hint: Don't forget to use the new solution's total volume.
0.208 M HCI
8.78 M
0.180 M
2.27 M

Inorganic Chemistry
Preparation and Properties of CompoundsDetermine the number of atoms that bear non-zero formal charges in the Lewis structure given below exactly as it is depicted.

Inorganic Chemistry
Preparation and Properties of CompoundsBy titration, it is found that 63.1 mL of 0.194 M NaOH(aq) is needed to neutralize 25.0 mL of HCl(aq). Calculate the concentration of the HCl solution.

Inorganic Chemistry
Preparation and Properties of CompoundsThe angle between the fluorines in PF3 are smaller than the angles between the fluorines in CF4, even thoughthey both have four pairs of electrons around the central atom. Explain why the fluorines are being pushed a little closer together in PF3 than they are in CF4

Inorganic Chemistry
Preparation and Properties of CompoundsBiphenyl, C₁2H10 , is a nonvolatile, nonionizing solute that is soluble in benzene, C6H6.
At 25 °C, the vapor pressure of pure benzene is 100.84 Torr. What is the vapor pressure of a solution made from dissolving 12.1 g of biphenyl in 33.9 g of benzene?

Inorganic Chemistry
Preparation and Properties of Compounds8. Balance the following redox reaction if it occurs in neutral solution. What are the coefficients in front of Ti²+ (aq) and Fe2+ (aq) in the balanced reaction?
Ti²+ (aq) +Fe³+ (aq) → Ti4+ (aq) + Fe²+ (aq)
D) 3 and 1
A) 1 and 2
B) 2 and 1
C) 2 and 2
E) 1 and 3

Inorganic Chemistry
Preparation and Properties of CompoundsHow many milliliters of 9.39 M nitric acid solution should be used to prepare 3.50 L of 0.300 M HNO3?

Inorganic Chemistry
Preparation and Properties of CompoundsIn the laboratory you dissolve 20.8 g of lead(II) nitrate in a volumetric flask and add water to a total volume of 375 mL.
What is the molarity of the solution?
What is the concentration of the lead cation?
What is the concentration of the nitrate anion?

Inorganic Chemistry
Preparation and Properties of CompoundsSolid Mass: 2.51 g
What is the volume of the solution, in liters?
amount added: 0.0129 mol

Inorganic Chemistry
Preparation and Properties of CompoundsWhen aqueous solutions of zinc iodide and potassium phosphate are combined, solid zinc phosphate and a solution of potassium iodide are formed. The net ionic equation for this reaction is:

Inorganic Chemistry
Preparation and Properties of CompoundsYou wish to make a 0.316 M perchloric acid solution from a stock solution of 6.00 M perchloric acid. How much concentrated acid must you add to obtain a total volume of 75.0 mL of the dilute solution?

Inorganic Chemistry
Preparation and Properties of CompoundsThe compound ammonium cyanide is a strong electrolyte. Write the reaction when solid ammonium cyanide is put into water.
Include states of matter in your answer.

Inorganic Chemistry
Preparation and Properties of CompoundsA chemist adds 275.0 mL of a 3.4M potassium iodide (KI) solution to a reaction flask. Calculate the mass in kilograms of potassium iodide the chemist has added to the flask. Round your answer to 2 significant digits.